The word osmosis is widely used in both science and everyday English, but its full meaning is often misunderstood. In biology and chemistry, osmosis is a fundamental process that explains how water, molecules, and solutions behave across a membrane.
In everyday language, people sometimes use the word “osmosis” to describe learning something naturally or absorbing ideas without effort. However, in science, osmosis has a very specific meaning tied to the movement of water across a semipermeable membrane due to differences in solute concentration.
Understanding this scientific definition helps connect what we observe in real life, such as plant growth, hydration, and even medical treatments, to the processes happening at the cellular level.
In this article, we will explore the osmosis meaning, how it works, and why it matters in both science and everyday life.

What Is Osmosis? 🔄
Osmosis is the process where water molecules move across a semipermeable membrane from an area of lower solute concentration to an area of higher solute concentration. If one side has a high concentration of solutes, there must be less water present. If, on the other side of the semi-permeable barrier, there is a lower concentration of solutes, thus a higher potential for water, water molecules will cross the barrier to make the concentration of solutes equal on both sides1.

This movement continues until the system reaches a balance, known as equilibrium, where the concentration of solutes is similar on both sides of the membrane. Even at this point, water molecules still move back and forth, but there is no overall or “net” movement in one direction. This constant adjustment helps maintain stable conditions within cells and ensures that important processes, such as nutrient transport and waste removal, can occur efficiently.
Jellyfish are 95% Water: If a jellyfish is placed in water with a different salt level, osmosis causes it to instantly shrink or swell.
In simpler terms, osmosis is how water naturally moves to even things out. Imagine two containers separated by a thin barrier. One side has more dissolved particles (like salt), and the other has less.
Water will move through the barrier toward the side with more particles to dilute it and create balance. Think of it like this: Water moves to where it is “needed” most.
How Does Osmosis Work? 💧
At the molecular level, osmosis happens because of differences in concentration.
Mechanism
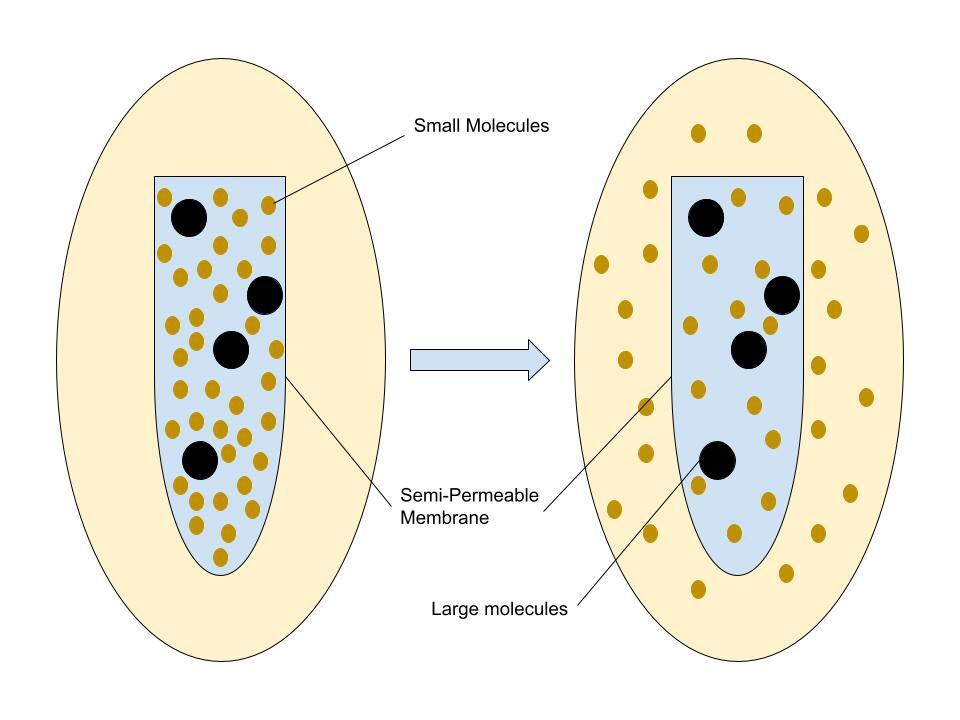
Water molecules are constantly moving. When there is a concentration gradient (an imbalance), water moves through a semipermeable membrane, which allows water to pass but blocks larger molecules.
The membrane acts like a filter:
- Water passes through
- Solutes (like salt or sugar) cannot
As a result, water flows toward the side with higher solute concentration until equilibrium is reached.
Factors Influencing Osmosis
Several factors affect how quickly and in which direction osmosis occurs:
Understanding these factors helps scientists control osmosis in real-world applications.
Osmosis in Biological Systems 🧪
Osmosis is essential for life. Every cell in every organism depends on it to function properly. It regulates the movement of water across the cell membrane, helping cells maintain the right balance between solute concentration inside and outside the cell.

This balance is critical because cells need a stable internal environment to carry out processes like nutrient absorption, waste removal, and energy production.
Cellular Processes
Inside the body, osmosis helps maintain balance between the inside and outside of cells by controlling how water moves across the semipermeable cell membrane. This movement is driven by differences in solute concentration between the cell (inside) and its surrounding solution (outside).
Because the membrane allows water to pass but not most solutes, water shifts to balance concentrations and maintain stable conditions inside the cell. Because the membrane allows water to pass but not most solutes, water shifts to balance concentrations and maintain stable conditions inside the cell. This selective permeability means that water can move freely through the cell membrane, while larger or charged solute molecules remain on their respective sides.
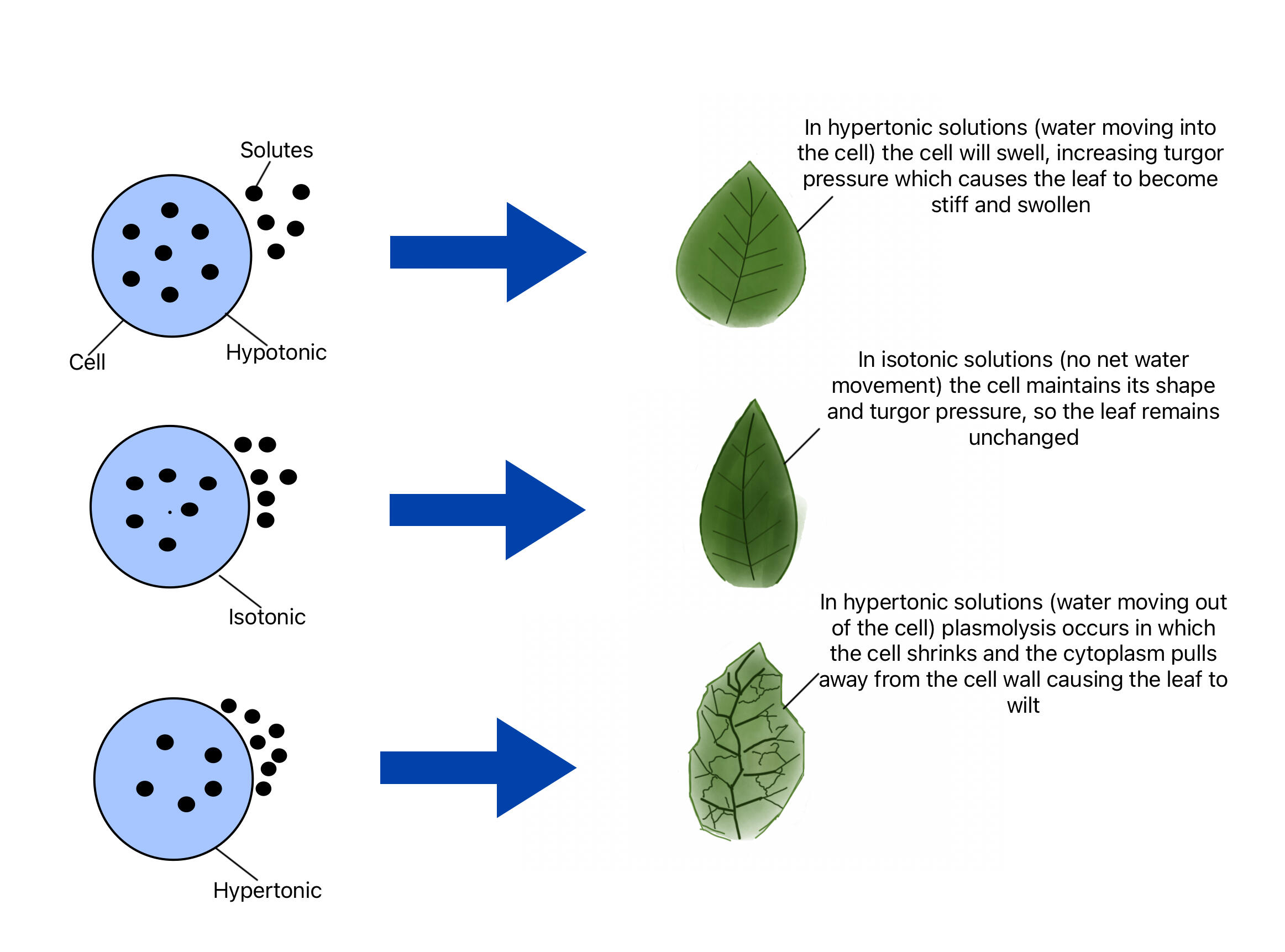
As a result, water moves toward the area with higher solute concentration to dilute it, helping equalize the solution on both sides. This movement supports homeostasis, ensuring that the cell maintains proper internal pressure, hydration, and conditions needed for processes like nutrient transport, enzyme activity, and waste removal2.
There are three important types of solutions:
Hypotonic solution
Lower solute concentration outside the cell → water enters the cell
Hypertonic solution
Higher solute concentration outside the cell → water leaves the cell
Isotonic solution
Equal solute concentration → no net movement of water
These conditions directly affect how a cell behaves. In a hypotonic environment, the net movement of water into the cell increases internal pressure, which may cause the cell to swell and potentially burst. In a hypertonic environment, water leaves the cell, reducing internal pressure and causing the cell to shrink or become dehydrated. In an isotonic environment, water moves in and out at equal rates, maintaining balance and allowing the cell to function normally.
It would help to know the function of cell structures to fully understand that concept, wouldn't it?
This constant regulation of water movement is essential for maintaining homeostasis, ensuring that cells keep their shape, pressure and internal conditions stable so they can carry out vital biological processes.
Examples in Organisms
These differ organism by organism!
Plants
In plants, osmosis helps roots absorb water from the soil because root cells have a higher solute concentration than the surrounding soil.
Water moves across the semipermeable membrane into the cells, filling the vacuole and creating turgor pressure.
This pressure pushes against the cell wall, keeping plants firm and upright. When plants lose water, turgor pressure decreases, causing them to wilt. Osmosis also helps move water through the plant, supporting nutrient transport, growth, and photosynthesis.

This continuous movement of water allows minerals from the soil to travel upward through the plant. It also helps maintain internal balance, ensuring that plant cells stay hydrated and function properly.
Humans and Animals
In the human body, osmosis is critical for maintaining balance within cells and across body systems. It regulates how water moves across cell membranes, helping cells stay properly hydrated and function effectively.
Osmosis plays an important role in:
Without osmosis, cells would not maintain proper balance, leading to disrupted homeostasis. This could affect everything from organ function to overall health, as cells rely on stable internal conditions to survive and perform essential biological processes.
Learn more about the structure of plant and animal cells that permit osmosis.

Practical Applications of Osmosis 💧🧠
Osmosis is not just a biological concept, it is used in many real-world technologies that rely on controlling how water, solutes, and solutions move across a semipermeable membrane. By understanding and manipulating osmotic pressure, scientists and engineers are able to solve problems related to clean water, health, and industry3.
Reverse Osmosis
One of the most important applications is reverse osmosis, a process that works against the natural direction of osmosis.

In normal osmosis, water moves toward a higher solute concentration. In reverse osmosis, external pressure is applied to force water in the opposite direction through a semipermeable membrane, leaving impurities behind.
In reverse osmosis:
- Pressure is applied to the solution
- Water is pushed through a membrane
- Dissolved salts, chemicals, and contaminants are removed
This process is widely used because it can separate very small particles from water, including substances that are not visible to the eye. Reverse osmosis is used for:
Drinking water purification 🚰
Removing contaminants and improving water quality.
Desalination 🧂🙅
Turning seawater into fresh water.
Industrial filtration 🏭
Cleaning water used in manufacturing and food production
Because of its ability to filter out extremely small molecules, reverse osmosis is one of the most effective water purification methods available today.
Medical Applications
Osmosis is also essential in healthcare, where maintaining the correct balance of water and solutes is critical for patient safety and overall body function. Medical treatments often depend on carefully controlling how fluids move across semipermeable membranes, especially at the level of cells and blood vessels.
One key example is dialysis, a life-saving treatment for people with kidney failure. When the kidneys are not able to properly filter waste, dialysis uses the principles of osmosis and diffusion to clean the blood.
In dialysis:
- A semipermeable membrane separates the patient’s blood from a special fluid called dialysate
- Waste products and excess solutes (like urea and salts) move out of the blood into the dialysate
- Clean fluid helps restore the correct balance of water and solutes in the body
Because of differences in solute concentration, water also moves across the membrane, helping remove excess fluid from the bloodstream. This process closely mimics the natural function of healthy kidneys, which regulate both waste and fluid levels to maintain homeostasis.
Osmosis is also important in several other medical contexts:
Overall, osmosis plays a vital role in healthcare by helping regulate fluid balance, protect cells, and support life-saving treatments. Without careful control of osmotic processes, many modern medical interventions would not be possible3.
Common Misconceptions About Osmosis ⚠️
Misconception 1: Osmosis is the same as diffusion
While osmosis and diffusion are related processes, they are not the same. Diffusion refers to the movement of any type of molecules (such as gases or solutes) from an area of high concentration to low concentration, and it does not always require a membrane.
Osmosis, on the other hand, is a very specific type of diffusion. It involves only water (the solvent) moving across a semipermeable membrane. The direction of movement is based on solute concentration: water moves toward the area with higher solute concentration to balance the solution.
In short:
- Diffusion = movement of many types of molecules
- Osmosis = movement of water only, through a membrane
There's lots more to diffusion: its purpose and usefulness in organisms, how it works in plants and animals, and which factors affect diffusion.
Misconception 2: Osmosis requires energy
Osmosis is a passive process, meaning it does not require energy from the cell. It happens naturally because of differences in concentration between two solutions.
Water molecules are constantly moving randomly. When there is a concentration gradient, more water moves in one direction than the other, creating a net movement. This continues until equilibrium is reached.
This movement is driven by differences in solute concentration on either side of a semipermeable membrane, which influences where water is more likely to move. Even though individual molecules move unpredictably, the overall pattern follows a clear direction toward balance.
This constant motion helps maintain stable conditions inside cells and supports important processes like nutrient transport and waste removal.
This is different from active transport, where cells must use energy (ATP) to move substances against a concentration gradient. Osmosis works “automatically” because of natural molecular motion.
Misconception 3: Movement stops completely
Even when a system reaches balance (equilibrium), water molecules do not stop moving. Instead, they continue to move in both directions across the membrane.
However, once equilibrium is reached, the movement becomes balanced:
- Water moves into the cell
- Water moves out of the cell at the same rate
This results in no net movement, even though molecular motion continues. This is called dynamic equilibrium, where movement is constant but balanced.
Why These Misconceptions Matter
Understanding these differences is important because it helps explain how cells maintain balance, how solutions behave, and how processes like osmosis, diffusion, and active transport work together in living systems.
When your fingers stay in water, osmosis allows water to interact with the outer layers of your skin. However, the wrinkly effect is mainly caused by your nervous system, which makes blood vessels shrink and the skin pucker. Wrinkles are not just from soaking: they’re a controlled body response that may help improve grip in wet conditions.
Clearing up these misconceptions makes it easier to understand more advanced concepts in biology and chemistry.
Find marine biology courses here on Superprof.
Osmosis is a fundamental process
Osmosis is a fundamental scientific process that explains how water, solutes, and solutions interact across membranes. From maintaining the health of a single cell to powering technologies like reverse osmosis, it plays a vital role in both biology and chemistry.
Understanding the osmosis meaning not only helps with science learning but also connects to real-world applications that affect our daily lives.
References
1. Encyclopaedia Britannica. (2026). Osmosis. https://www.britannica.com/science/osmosis
3. OpenStax. (2013). Concepts of Biology: Passive transport. Rice University. https://openstax.org/books/concepts-biology/pages/3-5-passive-transport
Summarize with AI:















